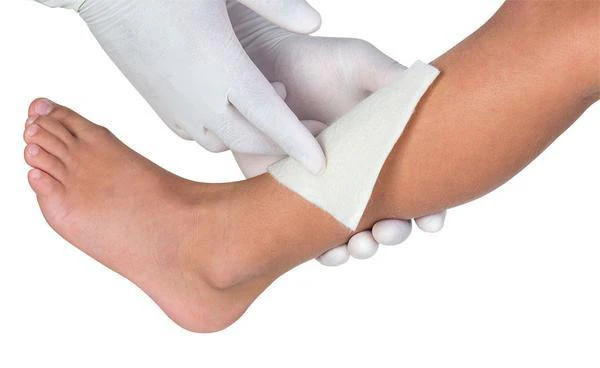
Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing - All Sizes
A soft-adherent TLC-NOSF matrix is combined with polyacrylate poly-absorbent fibres, which guarantee wound cleaning.
FEATURES:
- Healing properties
- Comfort and ease of use
BENEFITS:
- Reduction of healing time combined with cleaning (slough, exudate, debris)
- Absorption of exudate
- Management of minor bleeding wounds
- Creation and maintenance of a moist wound environment that promotes healing
- Atraumatic removal
- Respect of peri-wound skin
- Pain free removal
- Very conformable and easy to reposition
- A vapour permeable waterproof outer film with silicone adhesive on the edges
INDICATIONS:
UrgoStart® Plus NA is indicated for all stages (from the desloughing stage to complete healing) of:
- Exuding wounds
- chronic wounds
- leg ulcers
- pressure injuries
- diabetic foot ulcers
- long standing acute wounds.
CONTRAINDICATIONS:
- UrgoStart® Plus NA facilitates the management of minor bleeding wounds. However it should not be used for heavily bleeding wounds.
- In order not to delay any optimal treatment, UrgoStart® Plus NA is contra-indicated in cancerous wounds and wounds which may reveal a deep abscess.
- Do not use when there is a known sensitivity to UrgoStart® Plus NA.
PRECAUTIONS:
- The soft-adherent layer of UrgoStart® Plus NA adheres to latex surgical gloves. Therefore it is recommended that the tabs be used to facilitate application of the dressing.
- As it includes a super-absorbent layer, UrgoStart® Plus NA should not be cut.
- If the wound shows signs of local infection, it is recommended that anti-microbial treatment is used first before starting treatment with UrgoStart® Plus NA.
- If an atypical ulcer presents induration or over-granulation, treatment with UrgoStart® Plus NA should only be initiated after verifying that ulcer deterioration is absent, to prevent any delay in diagnosis.
- In the absence of clinical data regarding Epidermolysis Bullosa (even for longstanding lesions), the use of UrgoStart® Plus NA is not recommended.
- Stinging or even painful sensations may be reported at the start of the treatment. These are usually linked to the healing process and the need to suspend treatment is rare.
- During desloughing, the wound may appear to get larger due to the gradual elimination of slough.
- Concomitant use with a cream, an ointment, an emulsion is not recommended.
- UrgoStart® Plus NA must not be used in a hyperbaric chamber.
- Sterile individual packaging, for single use only: re-using a disposable dressing can lead to the risks of infection.
- Do not re-sterilise the dressing.
- (*) Check that the sterile protector is intact before use. Do not use if package is damaged.
| Product Code | Description |
| 100440 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 6cm x 6cm |
| 100441 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 10cm x 10cm |
| 100442 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 15cm x 20cm |
A soft-adherent TLC-NOSF matrix is combined with polyacrylate poly-absorbent fibres, which guarantee wound cleaning.
FEATURES:
- Healing properties
- Comfort and ease of use
BENEFITS:
- Reduction of healing time combined with cleaning (slough, exudate, debris)
- Absorption of exudate
- Management of minor bleeding wounds
- Creation and maintenance of a moist wound environment that promotes healing
- Atraumatic removal
- Respect of peri-wound skin
- Pain free removal
- Very conformable and easy to reposition
- A vapour permeable waterproof outer film with silicone adhesive on the edges
INDICATIONS:
UrgoStart® Plus NA is indicated for all stages (from the desloughing stage to complete healing) of:
- Exuding wounds
- chronic wounds
- leg ulcers
- pressure injuries
- diabetic foot ulcers
- long standing acute wounds.
CONTRAINDICATIONS:
- UrgoStart® Plus NA facilitates the management of minor bleeding wounds. However it should not be used for heavily bleeding wounds.
- In order not to delay any optimal treatment, UrgoStart® Plus NA is contra-indicated in cancerous wounds and wounds which may reveal a deep abscess.
- Do not use when there is a known sensitivity to UrgoStart® Plus NA.
PRECAUTIONS:
- The soft-adherent layer of UrgoStart® Plus NA adheres to latex surgical gloves. Therefore it is recommended that the tabs be used to facilitate application of the dressing.
- As it includes a super-absorbent layer, UrgoStart® Plus NA should not be cut.
- If the wound shows signs of local infection, it is recommended that anti-microbial treatment is used first before starting treatment with UrgoStart® Plus NA.
- If an atypical ulcer presents induration or over-granulation, treatment with UrgoStart® Plus NA should only be initiated after verifying that ulcer deterioration is absent, to prevent any delay in diagnosis.
- In the absence of clinical data regarding Epidermolysis Bullosa (even for longstanding lesions), the use of UrgoStart® Plus NA is not recommended.
- Stinging or even painful sensations may be reported at the start of the treatment. These are usually linked to the healing process and the need to suspend treatment is rare.
- During desloughing, the wound may appear to get larger due to the gradual elimination of slough.
- Concomitant use with a cream, an ointment, an emulsion is not recommended.
- UrgoStart® Plus NA must not be used in a hyperbaric chamber.
- Sterile individual packaging, for single use only: re-using a disposable dressing can lead to the risks of infection.
- Do not re-sterilise the dressing.
- (*) Check that the sterile protector is intact before use. Do not use if package is damaged.
| Product Code | Description |
| 100440 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 6cm x 6cm |
| 100441 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 10cm x 10cm |
| 100442 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 15cm x 20cm |
Original: $241.55
-65%$241.55
$84.54Description
A soft-adherent TLC-NOSF matrix is combined with polyacrylate poly-absorbent fibres, which guarantee wound cleaning.
FEATURES:
- Healing properties
- Comfort and ease of use
BENEFITS:
- Reduction of healing time combined with cleaning (slough, exudate, debris)
- Absorption of exudate
- Management of minor bleeding wounds
- Creation and maintenance of a moist wound environment that promotes healing
- Atraumatic removal
- Respect of peri-wound skin
- Pain free removal
- Very conformable and easy to reposition
- A vapour permeable waterproof outer film with silicone adhesive on the edges
INDICATIONS:
UrgoStart® Plus NA is indicated for all stages (from the desloughing stage to complete healing) of:
- Exuding wounds
- chronic wounds
- leg ulcers
- pressure injuries
- diabetic foot ulcers
- long standing acute wounds.
CONTRAINDICATIONS:
- UrgoStart® Plus NA facilitates the management of minor bleeding wounds. However it should not be used for heavily bleeding wounds.
- In order not to delay any optimal treatment, UrgoStart® Plus NA is contra-indicated in cancerous wounds and wounds which may reveal a deep abscess.
- Do not use when there is a known sensitivity to UrgoStart® Plus NA.
PRECAUTIONS:
- The soft-adherent layer of UrgoStart® Plus NA adheres to latex surgical gloves. Therefore it is recommended that the tabs be used to facilitate application of the dressing.
- As it includes a super-absorbent layer, UrgoStart® Plus NA should not be cut.
- If the wound shows signs of local infection, it is recommended that anti-microbial treatment is used first before starting treatment with UrgoStart® Plus NA.
- If an atypical ulcer presents induration or over-granulation, treatment with UrgoStart® Plus NA should only be initiated after verifying that ulcer deterioration is absent, to prevent any delay in diagnosis.
- In the absence of clinical data regarding Epidermolysis Bullosa (even for longstanding lesions), the use of UrgoStart® Plus NA is not recommended.
- Stinging or even painful sensations may be reported at the start of the treatment. These are usually linked to the healing process and the need to suspend treatment is rare.
- During desloughing, the wound may appear to get larger due to the gradual elimination of slough.
- Concomitant use with a cream, an ointment, an emulsion is not recommended.
- UrgoStart® Plus NA must not be used in a hyperbaric chamber.
- Sterile individual packaging, for single use only: re-using a disposable dressing can lead to the risks of infection.
- Do not re-sterilise the dressing.
- (*) Check that the sterile protector is intact before use. Do not use if package is damaged.
| Product Code | Description |
| 100440 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 6cm x 6cm |
| 100441 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 10cm x 10cm |
| 100442 | Urgostart Plus NA Lipido-Colloid Sucrose-Octasulfate Polyfibrous Dressing 15cm x 20cm |